Sustain / Real Bread Campaign / About / Marketing and labelling
Ascorbic acid
The Campaign has a simple, clear-cut and universally-accessible starting point for its definition of Real Bread: no additives.
As ascorbic acid (E300) is one commonly-used additive that some producers might ‘overlook’ when it is already in the flour they use (always read the label!), or excuse ‘because it’s only vitamin C’, we believe it deserves a closer look.
- Introduction
- Is ascorbic acid harmful?
- Isn’t ascorbic acid just vitamin C?
- How do I know if there’s ascorbic acid in flour or a loaf?
- What function does ascorbic acid perform in baking?
- How does it work?
- Where does ascorbic acid come from?
- Do the Campaign’s criteria allow orange or lemon juice?
- For how long has ascorbic acid been used in baking?
- More links
See also
Introduction
Just say no!
You really do not need to use E300, or any other additive, in your baking.
To be clear: ascorbic acid (aka E300 or l-ascorbic acid)* is an additive and a loaf made using it - either added to the flour by the miller or added to the dough by the baker - is not what the Campaign calls Real Bread.
Ultimately, it is up to each of us to decide what we want to put into the food we make and the food we put into our (and in some cases our children’s) mouths.
We hope that, like all other pages on our site, this makes a contribution to the information that helps you to make choices about the food you make and eat.
Some notes on ascorbic acid
- It is an unnecessary additive, which can weaken the gluten in longer-fermented doughs**
- One of its functions is to help ‘convey the impression of improved freshness to the customer’ (our italics)***
- We believe that bakers improving their knowledge and skills to get the most out of natural ingredients is more beneficial all round than falling back on an additive
- By helping gluten to ‘relax’ it can have the incidental effect of increasing the speed of rising, which is moving in the wrong direction of our aim to encouraging bakers to prolong dough fermentation
* Or using sodium ascorbate (aka E301 or sodium monoascorbate), a related chemical that can be used to perform the same function in baking.
** Ascorbic acid in long-fermented breads could: 'weaken the gluten structure with subsequent loss of gas retention in the dough. Ascorbic acid is thus best suited to no-time doughmaking systems.' Baking Problems Solved, p 55, S. Cauvain & L. Young, Woodhead Publishing (2001)
*** Idem p. 54
Is ascorbic harmful?
In general, the known health benefits of ascorbic acid far outweigh any potential risks, though please see the note below on the lack of health benefits of ascorbic acid as used in baking.
There is evidence that ascorbic acid can be harmful in high concentration or very large does (i.e. several grams a day), but as noted above, the amounts used in baking are so low, that this is not a consideration. Please read this in conjunction with the precautionary principle we believe should be applied to all food additives.
Links
Possible adverse health effects of vitamin C and ascorbic acid
Material Safety Data Sheet: Ascorbic acid
Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids
Isn’t ascorbic acid just vitamin C?
Consuming the ascorbic acid that can be used in baking does not provide the beneficial effects of vitamin C found in, say, an orange as it is:
- Used in far smaller quantities than the recommended daily intake
- Largely denatured (or degraded)* and its residues no longer have any beneficial properties of vitamin C
- A highly-refined substance, without the many complex bioflavonoids and other beneficial micronutrients that accompany vitamin C in fresh fruit
* Like many processing aids and other food additives, is it not 'destroyed' (as industrial loaf fabricators sometimes like to claim about such things) in the sense that its residues (and by-products) remain in the loaf.
How do I know if there’s ascorbic acid in flour or a loaf?
All bakers using The Real Bread Loaf Mark have signed an agreement that they will only use it for loaves baked without the use of any so-called processing aids or other addtives.
By law, any mill that has added ascorbic acid (E300) to its flour or baking mixes has to declare so on the label, so you will see it on the ingredients list.
The same applies to wrapped loaves and other pre-packed baked goods – though if deemed by the producer to be ‘processing aids’ some addtives would not have to appear on a loaf wrapper.
At present, bakers and retailers do not have to provide ingredients lists for food that is sold loose/unpackaged. As noted in our call for an Honest Crust Act, we demand a change in law to require that they do and in the meantime urge all bakers to do so voluntarily.
Of course, we also urge all bakers and millers currently using ascorbic acid to stop doing so!
What function does ascorbic acid perform in baking?
Industrial loaf fabricators categorise ascorbic acid as a flour ‘improver’ or dough conditioner. In the presence of oxygen, ascorbic acid becomes an oxidising agent and the ‘improvements’ to which it contributes include:
- Strengthened gluten
- Greater loaf volume
- Finer crumb (i.e. smaller, more even cells/bubbles)
- Increased tenderness of the crumb
- Reduced crust thickness
- Faster rising
‘The net result of AA is to improve the ability of dough to retain gas (as seen by increased oven spring) and to yield bread with a finer (smaller average cell size) crumb cell structure. These changes also result in bread crumb that is softer to the touch yet has the resiliency to recover much of its original shape after compression. This helps to convey the impression of proved freshness to the consumer.’ Baking Problems Solved, p 55, S. Cauvain & L. Young, Woodhead Publishing (2001)
How does it work?
Brace yourself for a bit of food science:
‘Ascorbic acid is, of course, a reducing agent but, during dough mixing, the enzyme ascorbic acid oxidase, naturally present in flour, catalyses its conversion to its oxidising form, dehydroascorbic acid (DHAA). In a reaction catalysed by another flour enzyme, DHAA converts glutathione (GSH), a tripeptide naturally found in wheat flour, to its dimer (GSSG). GSH, but not GSSG, can form disulfide bonds – so removing GSH prevents it from disrupting the disulfide crosslinking that otherwise form between gluten proteins.’
www.rsc.org/images/BreadChemistry
‘It is the network of disulfide bonds formed in the gluten structure that enables the dough to retain carbon dioxide produced by the yeast.’ Vitamin C in food processing
Where does ascorbic acid come from?
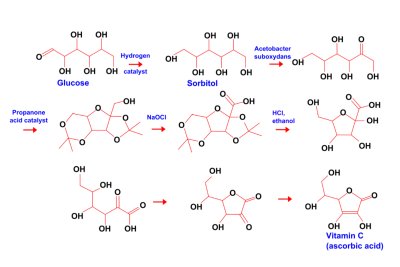
The ascorbic acid used as a food additive is not necessarily a highly-refined and purified extract from a natural vegetable source - e.g. citrus fruit. Often it is synthesised from sugars and other carbohydrates by a multi-step chemical and microbial process, e.g: 'Firstly, D-glucose is hydrogenated to D-sorbitol, followed by oxidation of the diacetone derivative of L-sorbose. The resulting diacetone-2-keto-l-gluconic acid is converted to L-ascorbic acid by heating with hydrochloric acid'
Unless the label states the source of ascorbic acid, the only way of knowing would be to ask its producer.
Do the Campaign’s criteria allow orange or lemon juice?
If you feel inclined to use a squeeze of lemon or orange juice in your dough for its ascorbic acid content, it would not fall foul of our Real Bread criteria.
Before you do, however, we do ask you to understand why and consider whether you really need to.
For how long has ascorbic acid been used in baking?
A UK patent for the use of ascorbic acid in baking was issued in 1936. It was, however, only when the tide turned against the use of other supposedly ‘safe’ chemicals on health grounds that ascorbic acid became more popular amongst industrial loaf fabricators, replacing the likes of potassium bromate and azodicarbonimide in the Chorleywood Process.
More links
ASCORBIC ACID….why use it?
Is L-Ascorbate the Same As Ascorbic Acid?
The Bioavailability of Different Forms of Vitamin C
Effect of Ascorbic Acid on the Rheological Properties of Wheat Fermented Dough
Functional role of ascorbic acid in bread-making
Substitution of ascorbic acid for potassium bromide in the making of French bread
Ascorbic Acid as an Oxidant in Wheat Flour Dough. II. Rheological effects
Functional role of ascorbic acid in bread-making
Vitamin C degradation during storage of fortified foods
Ascorbic acid
Real Bread Campaign: Finding and sharing ways to make bread better for us, our communities and planet.


